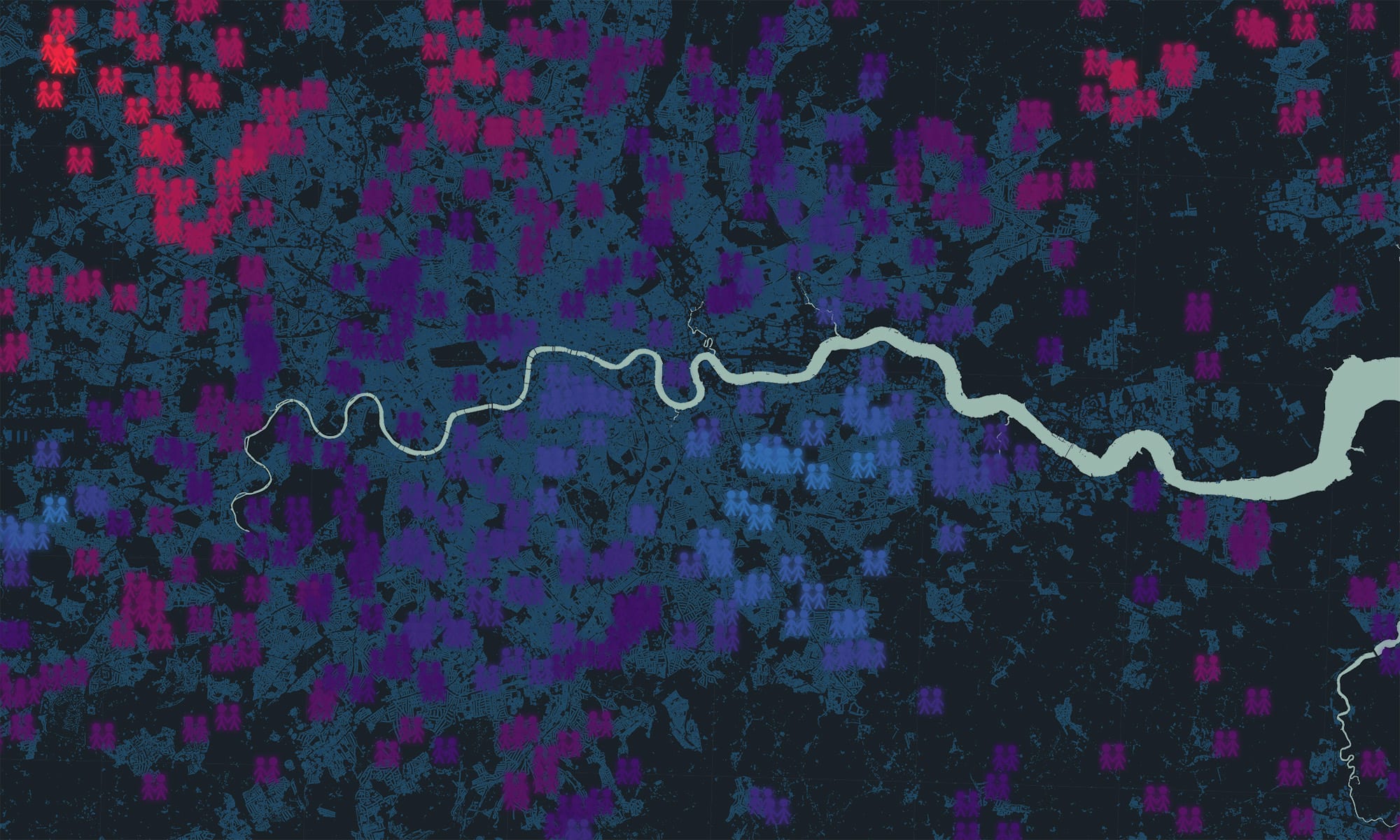
Twin studies allow us to estimate the relative contributions of nature and nurture to human phenotypes by comparing the resemblance of identical and fraternal twins. Variation in complex traits is a balance of genetic and environmental influences; these influences are typically estimated at a population level. But what if the balance of nature and nurture varies depending on where we grow up? We developed the spACE approach to analyse and map genetic and environmental hotspots using data from large twin cohorts.
EMBERS
The rapid evolution of genotyping and sequencing technologies means that genetic variation data are becoming readily available in the large populations necessary for research into the aetiology of complex traits and disorders. Now, rather than being limited by genotyping, we are starting to be restricted by the availability of phenotypic and environmental information. To understand the dynamics of genetic influences across development and in different contexts, we must develop new approaches that will complement traditional questionnaires and clinical data to give us affordable, repeatable and detailed assessments on a scale to match our vast repositories of genetic data.
TWIST
Genetic and environmental influences are dynamic. Our spACE project has shown how the importance of genes and environments can vary depending on where we grow up, and our longitudinal analyses have shown that for several traits heritability tends to increase with age. We wanted to take this a step further by exploring what happens to genetic and environmental influences when we experimentally change the environmental context through an intervention.
PhD studentship available
![]()
We have a PhD studentship available to start October 2017 as part of our EMBERS project, funded through the Medical Research Council (MRC) GW4 BioMed Doctoral Training Partnership (deadline 9.30am 8th June 2017):
Tracking dynamic genetic and environmental influences on mood in young adults through social media analysis
A fascinating finding from human genetics is that for many complex traits the balance of nature and nurture is not fixed, but can vary in response to different life stages or environments. This project will study how genetic and environmental influences on mood vary across emerging adulthood using high-resolution time course data from social media.
This project would suit a student who wishes to develop interdisciplinary skills in behavioural science, genetics and data science, using big data from social media and the human genome. You will be based at the MRC Integrative Epidemiology Unit at the University of Bristol, in the Dynamic Genetics lab.
Born around the same time as the commercial Internet, today’s emerging adults are the Internet generation, with most engaging frequently with their real-life peer groups through online social networks. For this generation, online social networks are integrated with offline networks, and are an important source of social support and interaction. If we are to understand social influences on mental health and disorder in this or future generations of adults, then we must take notice of online, as well as offline social activity. But although offline social networks are difficult to assess and track, online social networks are detailed databases of real-time social activity. Since social networks are an important factor in both positive and negative behaviours, with peer influence leading to outcomes such as depression, obesity and positive mental wellbeing, learning about these interactions is crucially important to our understanding of mental health and wellbeing.
The rapid evolution of genotyping and sequencing technologies means that genetic variation data are becoming readily available in the large populations necessary for research into the aetiology of complex traits and disorders. Now, rather than being limited by genotyping, we are starting to be restricted by the availability of phenotypic and environmental information. To understand the dynamics of genetic influences across development and in different contexts, we must develop new approaches that will complement traditional questionnaires and clinical data to give us affordable, repeatable and detailed assessments on a scale to match our vast repositories of genetic data.
Fortunately, new digital technologies can help us to do that. Our EMBERS (Emotion Monitoring by Electronic Remote Sensing) project uses online social networks and other electronic resources to collect high-resolution phenotypic and environmental data in genetically informative population samples. For example, we have collected over five million tweets from 2,500 participants in the Twins Early Development Study (TEDS). By comparing scores automatically coded from their tweets with standard questionnaire data collected at the same time, we have been able to establish the effectiveness of Twitter data for measuring positive and negative mood in emerging adulthood. This project will use these data to track the dynamics of genetic and environmental influences on positive and negative mood through this important life stage. The findings will help us to understand the complex aetiology of mental health and disorder, and how important influences evolve across time and in response to events.
You can apply for the studentship through the MRC GW4 BioMed DTP web site before 9.30am on Thursday 8th June 2017. For more information or an informal discussion before you apply, contact Oliver Davis or Claire Haworth.
A genome to call my own

A couple of months ago I signed up to send a tube full of saliva to a company called 23andMe. After seemingly endless spitting and six weeks of waiting, I received an email: my DNA report was ready.
My reason for signing up was a geeky fascination with the world of genetics. Ever since my first encounters with the subject at school, I have wanted to understand more about the incredible code of life lying within each of our cells and how this relates to human development, disease and behaviour. This fascination led me to be curious about my own hidden genetic liabilities.
What do the results reveal? Well, a lot of things I could have told you myself. I have high coffee metabolism. I have blue eyes and straight hair. However, some things are more enlightening; I now have an indication of my risk of 11 diseases including Alzheimer’s, breast cancer and Parkinson’s as well as my carrier status for over 40 single gene disorders. The ancestry report correctly reveals my grandparents’ genetic origins, telling me I am 23% Ashkenazi Jewish, my Grandfather’s contribution. And I am 3% Neanderthal (apparently most people fall between 2% and 4%). On top of this, 23andMe allow me access to the raw data for all of my genetic variants: perfect for a true genetics geek like me!
Beyond my simple curiosity, I had a second motivation. When I talk about my research with family and friends, I am increasingly aware of the common misconceptions about genetics. There is an overwhelming fear of genetic determinism: that “a gene for x” causes definite outcomes with nothing that can be done about it. This is very rarely true, and I wanted to see how 23andMe would handle such likely misinterpretation.
Some of the disease risk reports are for serious life-limiting conditions. However, increased risk of disease doesn’t necessarily mean the outcome is inevitable. For example, if you have high genetic risk of heart disease, you might make environmental changes to counteract your genetic propensity, such as regular exercise and a healthy diet. In the Dynamic Genetics lab we work towards understanding this complex interaction between environment and genetics. Nothing is necessarily set in stone, because genetics is about what is, not what could be.
So how does 23andMe do? I was impressed with the level of information provided for each disorder. Before viewing your risk for Alzheimer’s, breast cancer or Parkinson’s disease, you have to read information explaining the test results in order to unlock them. Text above each health report reads: “These reports show your results for specific genetic variants that are associated with a higher risk for developing certain health conditions. However, these reports cannot tell you whether you definitely will, or will not, develop the condition.”
But if I had received bad news, would the website provide enough support? It is very different to find these things out online as opposed to sitting opposite a genetic counsellor or your GP. And there will be many people who do not so much as glance at the supplementary information supplied with each disorder, but go straight to the results.
I was shocked by my own reaction to the report. It revealed I have 4 genetic variants associated with higher than average body mass index (BMI). Having spent my life jealous of my stick thin sister, my initial reaction was “this explains it!”. Although I am aware genes are not deterministic, I still surprised myself that I was much happier to place the blame with my genes rather than the fact I eat twice as much as her! (Of course, my eating behaviour is also influenced by my genes, but that’s a story for another day.) We somehow need to change this mind-set, to make a genetic liability for a negative outcome a motivator for positive change, rather than simply down-heartening. Especially when you consider that all of the currently known variants for BMI explain only around 2% of the variation between people (and I don’t even have the high risk version of all of them).
For many common disorders, our ability to predict risk from genetic data is just not very good right now. Complex disorders are typically highly polygenic (caused by many genetic variations, each of very small effect). The number of these variations that we know about is rapidly increasing with advances in technology and worldwide cooperation between research groups. However, we don’t yet know all of the genetic variants that are important, so estimates of genetic risk are currently based on incomplete information. And that’s even before you consider the many environmental factors from upbringing to lifestyle that also influence risk for complex disorders.
Having said this, the reports for traits and disorders included in 23andMe’s analysis did seem well thought out. For rare single gene disorders they report the causal variants, and for common disorders and complex traits they make it clear that the results are only predictions from what we know so far. One of the greatest challenges for human genetics today is understanding the genetic underpinnings of these complex traits. One important way to do this will be through large-scale research with genotype data from hundreds of thousands of participants. Some of this will be through publicly funded research like ours, and some will be through commercial enterprises like 23andMe; in July they announced their millionth customer.
So I am not alone. The number of people using personal genetic testing is increasing. It is hard not to interpret DNA as an explanation for your entire life and use it as an excuse. However, we know that behaviour is a complex thing. For most of the diseases and traits reported, genetics is only a part of the story and does not necessarily determine anything. As people become more informed about their genetics, they must also understand the importance of controlling their own environments. And on that note, I am off to the gym!
Computational behavioural genetics
https://www.youtube.com/watch?v=ONcbeK6FzcU
I was asked to give a fifteen-minute talk on Computational Behavioural Genetics at Europe’s first Computational Social Science conference at the University of Warwick last year. It was recorded for Warwick’s Big Data MOOC, and the organisers, Suzy Moat and Tobias Preis, have kindly allowed us to re-post it here. Thanks, guys!
Is happiness in our genes?
In this three-minute video, I talk about our Twins Wellbeing Intervention Study, TWIST. There’s more about TWIST, as well as my answers to the questions I raise here, as part of the University of Warwick’s What Makes Us Happy series.
Why nurture is just as important as nature for understanding genetics

We live in the age of the genome. Hardly a week goes by without a story about how genes influence our health or behaviour. There has been recent excitement around new advances in the genetics of schizophrenia, and genetic overlap between reading and maths. In the UK, the government is also pushing forward plans to map 100,000 genomes that will be matched to clinical data to drive “genomic medicine as part of routine care” in the NHS.
But genetic variation is only half the story. Environmental influences are important too, and we now know that our environments can interact with our genetic makeup, in ways that can be good and bad for our health.
One of the most striking findings from genetics research is that the influence of genes isn’t fixed. Even though our DNA sequence remains the same, the impact our genes have on us can alter with age and with the different environments we experience. Epigenetics, where the environment can change the expression of a gene without changing DNA, is only a small part of a whole field of science looking at changes in heritability due to interactions between genes and environment.
For example, we know that the importance of genetic influences for body weight increases as we get older. Genetic variation accounts for 48% of the differences between people in early childhood but by adolescence this rises to 78%. These estimated figures, from a study of thousands of twins in the Twins Early Development Study, have now been confirmed using analyses of DNA.
In fact, we see increasing heritability with age for many other human characteristics, such as IQ, where the importance of genes increases from 41% in early childhood up to 66% by young adulthood.
Drawing out genetic potential
One of the main mechanisms behind the increasing role of genetics as we get older is choice: we have more control over what we’re exposed to. We can choose whether to have a doughnut for lunch, whether to visit the library, or whether to cycle to work. These environments don’t just happen to us. To some extent we control, select and create our experiences and exposures. And because our genes can influence these choices too, we find ourselves in places and situations that in a sense draw out our genetic potential.
Our recent work tells us that the importance of genes and environments on childhood behaviour varies depending on where we grow up, shown in a series of UK maps of genetic and environmental influences for 45 childhood traits. For example, environmental influences were more important for disruptive classroom behaviour in London, compared to the rest of the UK.
The challenge now is to try and understand what in the environment can create these geographically distributed effects, because for this analysis at least, we know that genetic differences cannot explain these differing patterns.
The interplay between nature and nurture means that identifying which genes and which environments are having an effect is difficult; turning an already complex system, that links DNA with human behaviour, into a network of genetic and environmental pathways and intersections. But if we’re to understand the mechanisms behind these effects, and to develop ways of preventing disease or promoting better outcomes or behaviours we’ll have to get to grips with it.
One of the simpler examples is phenylketonuria, a disease where a defective variant of a gene means patients are not able to break down the amino acid phenylalanine, which builds up to toxic levels that affect brain development. Only by understanding the interaction between the gene and the presence of phenylalanine in our diets were researchers able to identify an effective treatment for a genetic disease: removing this amino acid from what affected children eat.
The diet is difficult to stick to, but it shows that it is sometimes possible to overcome genetic disease by changing the environment. More complex disorders that are influenced by many genes and many environments will of course need more complex interventions, and will probably have complicated mechanisms for us to unravel. But focusing on genetics alone means we will not fully understand these systems or processes.
Identifying which genes influence disease is important, but it’s just the first step. As we invest more in genetic research, we need to keep context in mind too. We should invest just as heavily in new methods for tracking and analysing behaviour, environments and health outcomes to the same degree of detail as we are now studying DNA. And that includes remaining open-minded about initiatives such as the NHS’s care.data, which would allow researchers access to more detailed information about our health outcomes.
The past 15 years have seen unprecedented and unexpected advances in genetic science. We should not underestimate the parallel advances we will make by understanding environmental influences and the way they interact with our genetic makeup. Nature and nurture are both important. We must be just as ambitious about understanding nurture as we are about understanding nature, because only by joining the two will we see the full picture.
![]()
This article was originally published on The Conversation. Read the original article.